|
The constitutive tyrosine phosphorylation of JAK3 was previously reported with other human T cell leukemia virus-I-infected cell lines ( 28). These data clearly demonstrate that IL-21 absolutely requires the γ c for its signal transduction. Similar results were obtained in EDγ-15, which is another cell line expressing the exogenous wild-type γ c (data not shown). Phosphorylation of JAK3 was detectable in these cell lines even before stimulation, and was significantly enhanced in EDγ-16 cells after stimulation with either IL-21 or IL-2 (Fig. IL-21 as well as IL-2 apparently induced phosphorylation of JAK1 in EDγ-16 cells but not in ED40515 − cells (Fig. We immunoprecipitated JAK1, JAK2, JAK3, and Tyk2 before and after IL-21 or IL-2 stimulation and then immunoblotted with P-Tyr-100. Hence, we examined tyrosine phosphorylation of the JAKs as a result of their activation. The IL-21R and the γ c were reported to associate with JAK1 and JAK3, respectively ( 19, 21, 25, 26, 27). All the cytokines whose receptors belong to the cytokine receptor family induce activation of JAKs with various combinations among the JAK family tyrosine kinases. Next, we examined whether IL-21 requires γ c for its intracellular signal transduction. However, the functional roles of the γ c are still not fully understood. Because the IL-15R complex is composed of the IL-2R β-chain and the γ c in addition to the unique IL-15R α-chain, dysfunction of IL-15 seems to lead to a severe defect of NK cells ( 16, 17). Gene disruption of the IL-2R β-chain as well as the γ c but not of IL-2, led to a defect of NK cell development ( 14, 15). Therefore, IL-7 was thought to be an essential cytokine for T and B cell differentiation and proliferation in mice. Gene disruption of either IL-7 or the IL-7R α-chain led to severe developmental defects of T and B cells ( 12, 13). These findings raised the next question of which cytokines are responsible for each hemopoietic cell development disturbed in XSCID patients. Furthermore, γ c gene-targeting mice demonstrated that various phenotypes of XSCID patients are caused by the dysfunction of the γ c ( 9, 10, 11). The mutant γ cs detected in the XSCID patients were revealed to have no ability for formation of the functional receptor complexes ( 8). The human γ c gene is located on the X chromosome, and patients with X-linked SCID (XSCID), a disease characterized by an absence of T and NK cells with a presence of nonfunctional B cells, have mutations in the γ c gene ( 6, 7). 
The IL-2R γ-chain was later uncovered to be an indispensable subunit of the IL-4, IL-7, IL-9, and IL-15 receptors and is now referred to as the common γ-chain (γ c) 3 ( 5). 
We previously reported the cloning of an IL-2R γ-chain and demonstrated it to be an essential component for the functional IL-2R complex ( 3, 4). Glycoprotein 130, which was originally discovered as a component of the IL-6R complex, is shared with IL-11, oncostatin M, LIF, ciliary neurotrophic factor, and cardiotrophin-1 ( 2). The common β-chain, which was originally identified as a GM-CSF receptor β-chain, is shared with IL-3 and IL-5 ( 1). View Moreĭivision of Structural Biology, Wellcome Trust Centre for Human Genetics, University of Oxford, Roosevelt Drive, Oxford OX3 7BN, UK.The most unique feature of the cytokine receptor family is their ability for sharing with many cytokines. These results offer new insights into the signalling mechanisms of pentameric ligand-gated ion channels and enhance current understanding of GABAergic neurotransmission. 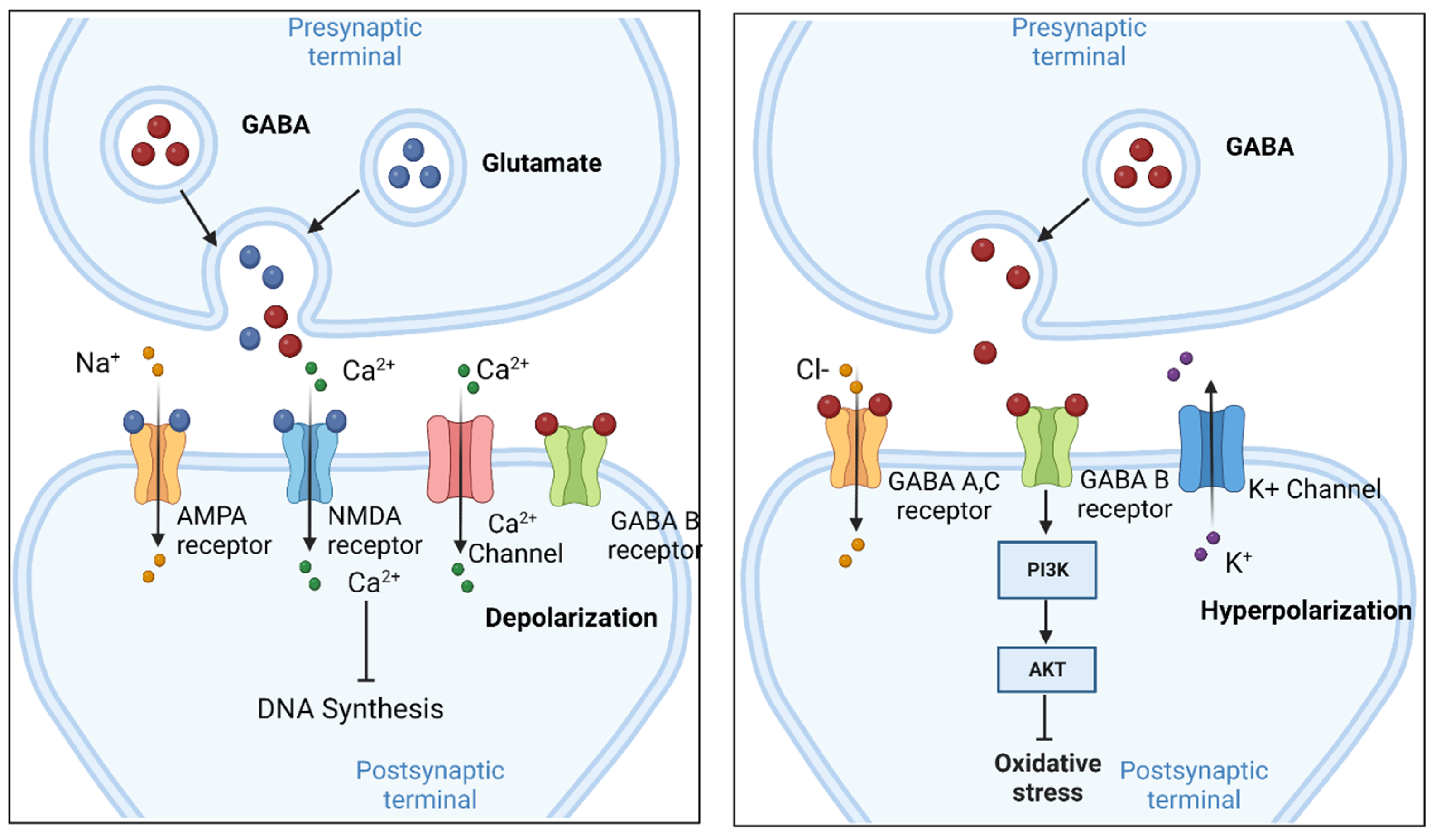
The channel region forms a closed gate at the base of the pore, representative of a desensitized state. 
The receptor was crystallized bound to a previously unknown agonist, benzamidine, opening a new avenue for the rational design of GABAAR modulators. This structure reveals architectural elements unique to eukaryotic Cys-loop receptors, explains the mechanistic consequences of multiple human disease mutations and shows an unexpected structural role for a conserved N-linked glycan. Here we present the first three-dimensional structure of a GABAAR, the human β3 homopentamer, at 3 Å resolution. A decline in GABAAR signalling triggers hyperactive neurological disorders such as insomnia, anxiety and epilepsy. Type-A γ-aminobutyric acid receptors (GABAARs) are the principal mediators of rapid inhibitory synaptic transmission in the human brain.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
